Perusing the internet, I came upon web pages detailing the use of didymium glass filters to do this very trick. Glassblowers have been using the glass in safety glasses for some time so that they can see the melting glass they're working on despite the sodium light of the flame they're working with. Photographers (as contrasted with astrophotographers) use didymium filters to boost the color saturation of the red end of the spectrum (as compared to the more yellowish middle) in landscape photography.
I found several photographers who claimed the Hoya filter to be the best, so I ordered one for my Nikon:

The filter is supposed to knock down the yellow of sodium outdoor lights in particular. After some additional internet skulduggery I found out more about skyglow & filter specifications Here is a compilation of a spectrum from sodium lights & the didymium filter's transmission performance:
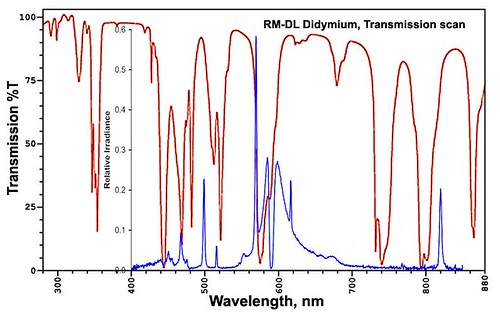
The blue line is the light pollution relative brightness, while the red line is the transmission curve for the filter. The higher the blue line, the brighter the sodium light is for that wavelength. The lower the red line, the less of that color light gets through the filter. So what I want is the "valleys" in the red line to cover up as much of the "peaks" in the blue line as is possible. I carefully aligned the wavelengths (color) for the 2 graphs so that they're as accurate as humanly possible.
You can see that the dip in the red line just below 600nm takes out a bunch of the peaks of the blue line there. It's not perfect, but every little bit helps. The sodium lines that I particularly was interested in is a pair at 589.0 & 589.6nm. This pair of lines is very familiar to chemists & astronomers alike, they're very distinctive.
The blue spectrum line, by the way, is courtesy testone22 and can be found here:
testone22's page in a new window.
And the red line is courtesy of Starna Cells, Inc. a Califormia manufacturer of spectrophotometer cells:
Starna's graphic in a new window.
Starna's home page in a new window.
He informs me that the actual peak has been inverted because of self-absorption (the discovery of which netted Kirchoff the 1862 Rumford medal). Thus the worst part of the light pollution has cancelled itself out! How convenient. Nonetheless, there is ample yellow light on either side of there from the pressure broadening in the high pressure lamps that is coloring the sky and a goodly part of this light is taken out by the filter.
By the way, here's a source for more didymium information:
Wikipedia's didymium page in a new window.
Wikipedia's didymium page in a new window.
Where I came on this fascinating tidbit:
"During World War I, didymium glass was reputedly used to send Morse Code across the battlefields. Didymium did not absorb enough light to make the variation in lamp intensity obvious, but anyone with binoculars attached to a prism could see the absorption bands flash on or off."
Wow! Using the unique spectral signature of the glass to hide the fact that Morse Code was being sent! Triksy hobbitses!
After seemingly innumerable days of clouds & rain (the "new astronomy gear curse"), it was clear enough last night to do an experiment. I quickly drew a little grid on a post-it note with my plan for exposures: 2 min, 4 min & 10 minute, both with & without the filter.
I headed outside with the camera on my fluid head tripod ready for use. I plunked down the camera and carefully framed the shot so that a neighbor's mercury-vapor "security" (aka "the burglar's friend") light was behind a small tree trunk and started a 2 minute exposure. I used the stopwatch feature of my iPod Touch to time the shot. I would have put red plastic over the bright screen but that prevents the touch control feature from working. I vowed to buy a simple stop watch like I used to have back when I worked in TV news.
So far so good, one exposure in the bag, now for a 4 minute shot...
Halfway through this shot, a motion control burglar assistance light came on in another location, off the frame to the right. I moved around to the front of the camera so my shadow was being cast on the lens and thought about what to do. I knew that this new source of light pollution was lighting up any close-by dust in the sky as well as the foreground with a different lighting regimen than the 2 minute shot had, so I decided that this image wouldn't work for me. You may have noticed a certain negative attitude toward outdoor lighting at night. I'll have to work up a blog post about the issues surrounding light pollution & light trespass. Stay tuned!
As the 4 minutes was up then anyway, I ended the shot as I normally would have and thought about my new options. Should I wait out the light and resume the exposure series I had planned on? I decided to press on and get a next shot, the 10 minute one, with me stuck standing in front (but not in the shot) to shade the lens.
I started the exposure and after a couple minutes I had a rude awakening.
Yet another motion controlled light came on! Aagh! This time it's a light that is supposed to scare away deer from a different neighbor's wine grapes. The deer are completely used to the light now and ignore it, but now I had a second source of light pollution that wasn't in either the 2 minute or 4 minute exposures! Worse of all, it was on-camera! I terminated the 10 minute exposure at 3 minutes or so.
Now I'm in a pickle. I have no good options about camera placement and no way to block all 3 lights that were shining in my face. I briefly considered getting out my stepladder and unscrewing the 2 offending lights when one after the other they both went out. Yay! I quickly decided to just get two 4 minute long exposures, one with and one without the filter. I held my breath while the camera's shutter was open and hoped for the best.
Okay, I didn't really hold my breath, but I was nervous, I'll tell you!
Success! Have a look:

You can easily see the difference between the non-filtered image and the filtered one. The annoying orange skyglow is not so orange any more and the contrast in significantly improved. It doesn't look like a dark sky site all of a sudden, but it is a definite improvement!
My next step in this saga is to make the short trek to McCormick and see what trouble I can get into up there. I'll have to take along my trusty Mac laptop so I can check up on my progress and respond to changes. Maybe Charlottesville will have an unexpected blackout and I can get the real thing!
Yeah, like THAT's ever gonna happen!

You can easily see the difference between the non-filtered image and the filtered one. The annoying orange skyglow is not so orange any more and the contrast in significantly improved. It doesn't look like a dark sky site all of a sudden, but it is a definite improvement!
My next step in this saga is to make the short trek to McCormick and see what trouble I can get into up there. I'll have to take along my trusty Mac laptop so I can check up on my progress and respond to changes. Maybe Charlottesville will have an unexpected blackout and I can get the real thing!
Yeah, like THAT's ever gonna happen!